Research
We study various research themes in synthetic molecular inorganic and organometallic chemistry, and we work on discovering fundamentally new compound classes including those with elements in rare oxidation states, unusually bonded fragments and rare and reactive metal hydride species. These generally show novel structures, have unseen properties and, as a consequence, often show a unique reactivity. We use these for applications in chemical synthesis, catalysis and other technologies. In our studies, we use complexes with elements from across the periodic table with a strong focus on main group elements. Many main group elements are earth abundant, non-toxic and even biocompatible. For example, the eight most abundant elements in earth’s crust account for approximately 98-99% of its mass, and seven of these are main group elements. Between them, they offer a wide range of properties, ionic or covalent radii, and accessible oxidation states. In our research, we employ and design a wide range of sterically demanding neutral and anionic ligands. For manipulations of air and moisture sensitive compounds, we routinely apply specialized synthetic Schlenk line and glove box techniques in our laboratories at the University of St Andrews. The main characterization methods we use to study these systems include multi-nuclear NMR spectroscopy and single crystal X-ray diffraction techniques among numerous other methods.
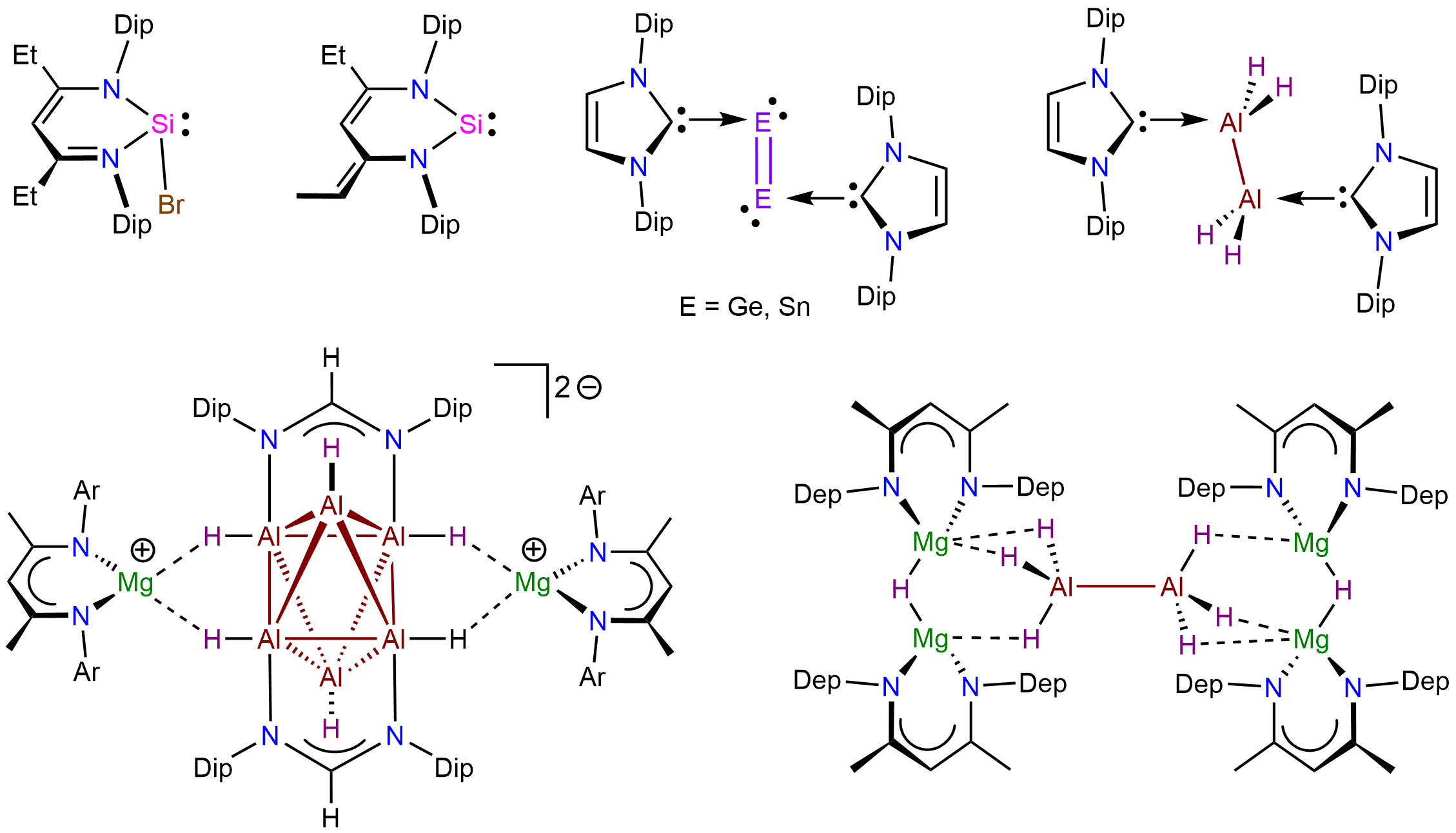
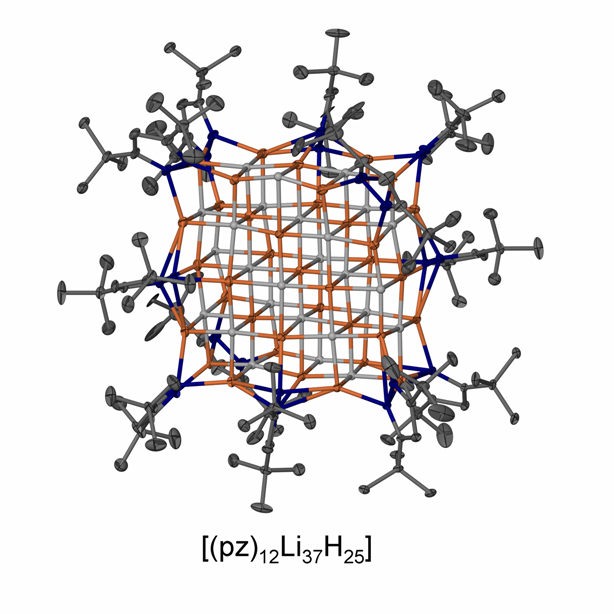
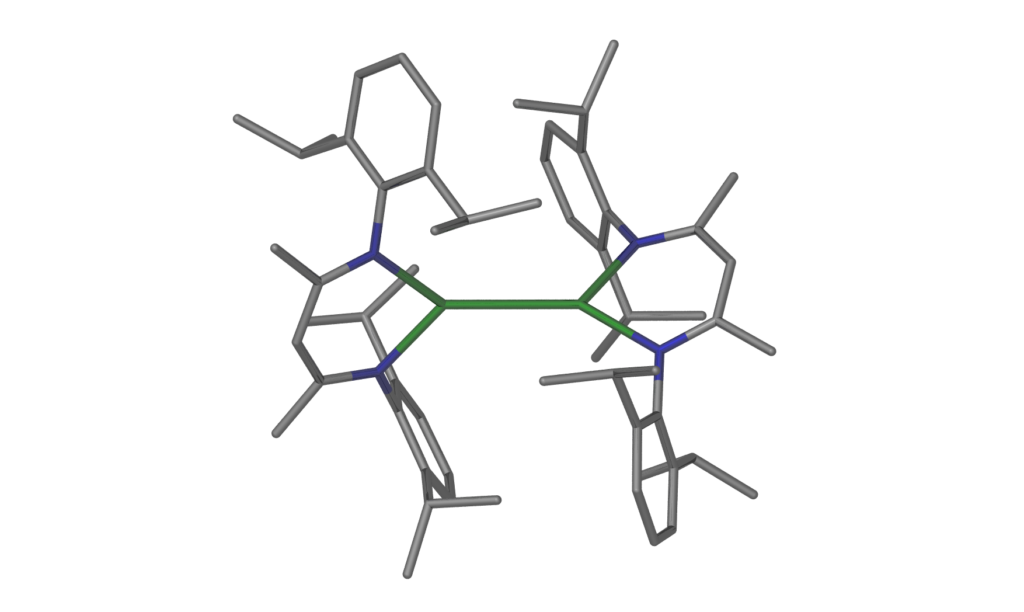
Unusual main group molecules and their reactivity – low oxidation state species, low coordinate species, donor-stabilized small molecules, element hydride complexes, rare organometallics, fulleride complexes and compounds with rare bonding modes
We study the chemistry of unusual molecules of the s- and p-block elements including compounds with elements in rare oxidation states and in novel bonding situations. These advances are enabled and controlled by employing suitable sterically demanding ligand systems. Selected examples of compound classes and reaction products we work with can be found in the images. Please visit the selected publications page for further information on recent work.